 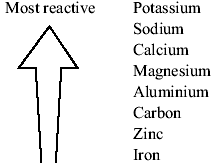
On the basis of reactions identify the most reactive metalsħ For doing displacement reactions in the laboratory, the form in which Zn metal is available is 
To show that zinc is more reactive than copper the correct procedure isĪ) Prepare CuSO 4 soln and dip Zn strip in it.Ħ. Can we store ZnSO 4 in aluminium container and why?Ī) Yes, because aluminium is less reactive than Zinc.ī) No, because aluminium is more reactive than Zinc.ĥ. After one hour the colour of the solution was observed to change fromģ. A piece of granulated Zinc was added to 5 mL of freshly prepared CuSO 4 solution kept in a test tube. Out of the following pairs of metallic salts, the pair of salts whose each salt forms coloured solution when dissolved separately in two beakers containing water isĢ. 
This type of prevention of corrosion is known as sacrificial protection as Zn sacrifices itself to protect Fe from rusting. Zn being more reactive will not allow hull to undergo rusting. (Ans: This is done in order to prevent rusting of hull of the ship. Write balanced chemical reaction involved. Zinc blocks are welded to the hulls of ships. This type of prevention of corrosion is known as sacrificial protection as Mg sacrifices itself to protect Fe from rusting. Mg being more reactive will not allow railway track to undergo rusting. (Ans: This is done in order to prevent rusting of Railway tracks. Railway tracks are connected to Mg or Zn metal after a particular distance. Write balanced chemical reaction involved.Īns: No, Zn will displace Cu: (Problems: - Precious copper sulphates solution will be lost in two ways: (i) Zn reacts with it and (ii) there will be holes developed on Zn due to the displacement reaction between Cu and Zn)ģ. Will you store copper sulphate solution in a zinc vessel? Give reasons to your answer. What happens when (Write balanced chemical equations for the reaction involved):Ģ. The metal pieces should be free from rust (oxide coating) on their surface.ġ. Use concentrated solutions of copper sulphate, magnesium sulphate and ferrous sulphate.Ģ. Therefore the reactivity order is: Cu < Fe < Zn < Alġ. Out of the four metals tested, Aluminium is the strongest and Copper the weakest.ģ. From the above experiment it is proved that stronger metals displace weaker metals from their compounds.Ģ. Write your comments/conclusions in the inference box.ġ. Record your observation in the following table.Ħ. Wait for about 25-30 minutes and observe the change that takes place in each test tube.ĥ. Repeat the experiment with Al 2(SO 4) 3, ZnSO 4 and FeSO 4 solutions as well.Ĥ. Take 5 ml (each) of copper sulphate solution in four different test tubes and put an iron nail to first test, a small copper wire to the second little aluminium powder to the third and a small zinc granule to the fourth. Prepare about 25 ml solutions of copper sulphate, ferrous sulphate aluminium sulphate and zinc sulphate.Ģ. Requirements:Test tubes, CuSO 4, FeSO 4, Al 2(SO 4) 3, ZnSO 4, Copper wire, Iron nail, zinc granules, Al powder, distilled water, test tube rack, beakers (100 ml) etc.ġ. Principle: More reactive metals displace less reactive metals from their compounds. Objective:To study the interaction of metals namely copper, iron, zinc and Aluminium with their salt solutions and arrange them according to their reactivity. STUDY OF DISPLACEMENT REACTION & REACTIVITY SERIES 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
